产品详情:
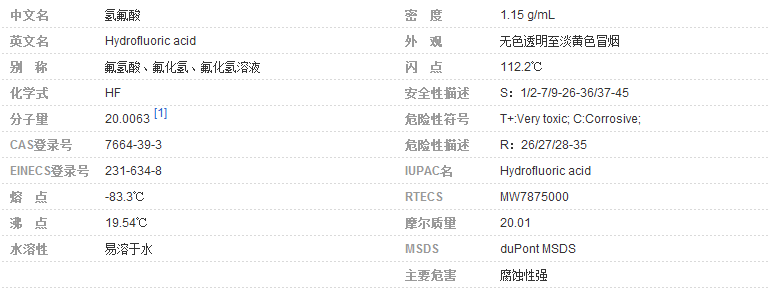
氢氟酸是氟化氢气体的水溶液,清澈,无色、发烟的腐蚀性液体,有剧烈刺激性气味。熔点-83.3℃,沸点19.54,闪点112.2℃,密度1.15g/cm³。易溶于水、乙醇,微溶于乙醚。因为氢原子和氟原子间结合的能力相对较强,使得氢氟酸在水中不能完全电离,所以理论上低浓度的氢氟酸是一种弱酸。具有极强的腐蚀性,能强烈地腐蚀金属、玻璃和含硅的物体。如吸入蒸气或接触皮肤会造成难以治愈的灼伤。实验室一般用萤石(主要成分为氟化钙)和浓硫酸来制取,需要密封在塑料瓶中,并保存于阴凉处。
需要购买的客户欢迎来电咨询:18015828251 QQ:2488095996